Direct interaction of SRY-related protein SOX9 and steroidogenic factor 1 regulates transcription of the human anti-Müllerian hormone gene
- PMID: 9774680
- PMCID: PMC109250
- DOI: 10.1128/MCB.18.11.6653
Direct interaction of SRY-related protein SOX9 and steroidogenic factor 1 regulates transcription of the human anti-Müllerian hormone gene
Abstract
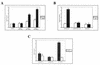
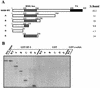
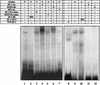
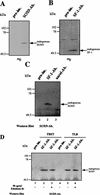
For proper male sexual differentiation, anti-Müllerian hormone (AMH) must be tightly regulated during embryonic development to promote regression of the Müllerian duct. However, the molecular mechanisms specifying the onset of AMH in male mammals are not yet clearly defined. A DNA-binding element for the steroidogenic factor 1 (SF-1), a member of the orphan nuclear receptor family, located in the AMH proximal promoter has recently been characterized and demonstrated as being essential for AMH gene activation. However, the requirement for a specific promoter environment for SF-1 activation as well as the presence of conserved cis DNA-binding elements in the AMH promoter suggest that SF-1 is a member of a combinatorial protein-protein and protein-DNA complex. In this study, we demonstrate that the canonical SOX-binding site within the human AMH proximal promoter can bind the transcription factor SOX9, a Sertoli cell factor closely associated with Sertoli cell differentiation and AMH expression. Transfection studies with COS-7 cells revealed that SOX9 can cooperate with SF-1 in this activation process. In vitro and in vivo protein-binding studies indicate that SOX9 and SF-1 interact directly via the SOX9 DNA-binding domain and the SF-1 C-terminal region, respectively. We propose that the two transcription factors SOX9 and SF-1 could both be involved in the expression of the AMH gene, in part as a result of their respective binding to the AMH promoter and in part because of their ability to interact with each other. Our work thus identifies SOX9 as an interaction partner of SF-1 that could be involved in the Sertoli cell-specific expression of AMH during embryogenesis.
Figures




References
-
- Bell D M, Leung K K H, Wheatley S C, Ng L J, Zhou S, Ling K W, Sham M H, Koopman P, Tam P P L, Cheah K S E. SOX9 directly regulates the type-II collagen gene. Nat Genet. 1997;16:174–178. - PubMed
-
- Carré-Eusèbe D, Di Clemente N, Rey R, Pieau C, Vigier B, Josso N, Picard J-Y. Cloning and expression of the chick anti-Müllerian hormone gene. J Biol Chem. 1996;271:4798–4804. - PubMed
-
- Clépet C, Schafer A J, Sinclair A H, Palmer M S, Lovell-Badge R, Goodfellow P N. The human SRY transcript. Hum Mol Genet. 1993;2:2007–2012. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
