The major outer membrane protein of Chlamydia psittaci functions as a porin-like ion channel
- PMID: 9784523
- PMCID: PMC108649
- DOI: 10.1128/IAI.66.11.5202-5207.1998
The major outer membrane protein of Chlamydia psittaci functions as a porin-like ion channel
Abstract
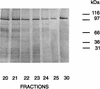
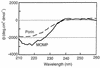
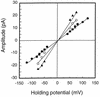
The major outer membrane protein (MOMP) of Chlamydia species shares several biochemical properties with classical porin proteins. Secondary structure analysis by circular dichroism now reveals that MOMP purified from Chlamydia psittaci has a predominantly beta-sheet content (62%), which is also typical of bacterial porins. Can MOMP form functional ion channels? To directly test the "porin channel" hypothesis at the molecular level, the MOMP was reconstituted into planar lipid bilayers, where it gave rise to multibarreled channels, probably trimers, which were modified by an anti-MOMP monoclonal antibody. These observations are consistent with the well-characterized homo-oligomeric nature of MOMP previously revealed by biochemical analysis and with the triple-barreled behavior of other porins. MOMP channels were weakly anion selective (PCl/PK approximately 2) and permeable to ATP. They may therefore be a route by which Chlamydia can take advantage of host nucleoside triphosphates and explain why some anti-MOMP antibodies neutralize infection. These findings have broad implications on the search for an effective chlamydial vaccine to control the significant human and animal diseases caused by these organisms.
Figures



References
-
- Batteiger B E, Rank R G, Bavoil P M, Soderberg L S. Partial protection against genital reinfection by immunization of guinea pigs with isolated outer membrane proteins of the chlamydial agent of guinea pig inclusion conjunctivitis. J Gen Microbiol. 1993;139:2965–2972. - PubMed
-
- Buzoni-Gatel D, Bernard F, Andersen A, Rodolakis A. Protective effect of polyclonal and monoclonal antibodies against abortion in mice infected by Chlamydia psittaci. Vaccine. 1990;8:342–346. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

