Role of gamma delta T cells in immunopathology of pulmonary Mycobacterium avium infection in mice
- PMID: 9784564
- PMCID: PMC108690
- DOI: 10.1128/IAI.66.11.5508-5514.1998
Role of gamma delta T cells in immunopathology of pulmonary Mycobacterium avium infection in mice
Abstract
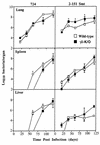
Several studies have shown that gamma delta T cells influence granuloma development after infection with intracellular pathogens. The role of gamma delta T cells in controlling the influx of inflammatory cells into the lung after Mycobacterium avium infection was therefore examined with gene-disrupted mice (K/O). The mice were infected with either M. avium 724, a progressively replicating highly virulent strain of M. avium, or with M. avium 2-151 SmT, a virulent strain that induces a chronic infection. gamma delta-K/O mice infected with M. avium 2-151 SmT showed early enhanced bacterial growth within the lung compared to the wild-type mice, although granuloma formation was similar in both strains. gamma delta-K/O mice infected with M. avium 724 showed identical bacterial growth within the lung compared to the wild-type mice, but they developed more-compact lymphocytic granulomas and did not show the extensive neutrophil influx and widespread tissue necrosis seen in wild-type mice. These data support the hypothesis that isolates of M. avium that induce protective T-cell-specific immunity are largely unaffected by the absence of gammadelta T cells. Whereas with bacterial strains that induce poor protective immunity, the absence of gamma delta T cells led to significant reductions in both the influx of neutrophils and tissue damage within the lungs of infected mice.
Figures




References
-
- Benson, C. A. 1994. Disease due to the Mycobacterium avium complex in patients with AIDS: epidemiology and clinical syndrome. Clin. Infect. Dis. 18(Suppl. 3):S218–S222. - PubMed
-
- Boismenu R, Havran W L. An innate view of γδ T cells. Curr Opin Immunol. 1997;9:57–63. - PubMed
-
- Boom W. The role of T-cell subsets in Mycobacterium tuberculosis infection. Infect Agents Dis. 1996;5:73–81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

