Ceramide inhibits inwardly rectifying K+ currents via a Ras- and Raf-1-dependent pathway in cultured oligodendrocytes
- PMID: 9786978
- PMCID: PMC6793552
- DOI: 10.1523/JNEUROSCI.18-21-08712.1998
Ceramide inhibits inwardly rectifying K+ currents via a Ras- and Raf-1-dependent pathway in cultured oligodendrocytes
Abstract
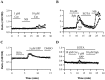
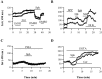
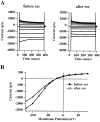
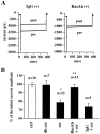
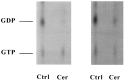
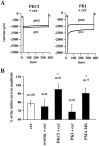
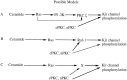
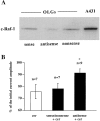
Ceramide is a lipid mediator implicated in apoptosis induced by proinflammatory cytokines in many cell types, including oligodendrocytes (OLGs). To determine whether ceramide modulates transmembrane signaling events in OLGs, we studied its effect on intracellular Ca2+ (Cai), resting membrane potential and inwardly rectifying K+ currents (IKir) in cultured neonatal rat OLGs. We report here that (1) exposure to C2-ceramide (cer) rarely increases OLG Cai, whereas sphingosine elicits sustained increase in Cai; (2) cer causes OLG depolarization, an effect mimicked by sphingosine-1-phosphate but not by sphingosine; and (3) cer, but not its inactive analog dihydroceramide, inhibits OLG IKir. The cer effect is attenuated by Ras antibody Y13-259, by protein kinase C inhibitory peptide (19-36), and by suppression of c-Raf-1 expression with antisense raf-1 oligonucleotides. We conclude that cer-induced OLG depolarization is mediated via inhibition of IKir by a Ras- and raf-1-dependent pathway, which results in the phosphorylation of the inward rectifier K+ channel protein.
Figures
Similar articles
-
Regulation of mitogen-activated protein kinases by sphingolipid products in oligodendrocytes.J Neurosci. 1999 Sep 1;19(17):7458-67. doi: 10.1523/JNEUROSCI.19-17-07458.1999. J Neurosci. 1999. PMID: 10460252 Free PMC article.
-
Ceramide-dependent regulation of p42/p44 mitogen-activated protein kinase and c-Jun N-terminal-directed protein kinase in cultured airway smooth muscle cells.Cell Signal. 2000 Dec;12(11-12):737-43. doi: 10.1016/s0898-6568(00)00119-4. Cell Signal. 2000. PMID: 11152959
-
Analysis of the K+ current profile of mature rat oligodendrocytes in situ.J Membr Biol. 2002 Oct 1;189(3):201-12. doi: 10.1007/s00232-002-1014-8. J Membr Biol. 2002. PMID: 12395285
-
Expression and modulation of K+ currents in oligodendrocytes: possible role in myelinogenesis.Dev Neurosci. 1989;11(2):118-31. doi: 10.1159/000111893. Dev Neurosci. 1989. PMID: 2663420 Review.
-
Signal transduction pathways in oligodendrocytes: role of tumor necrosis factor-alpha.Int J Dev Neurosci. 1995 Jun-Jul;13(3-4):351-67. doi: 10.1016/0736-5748(95)00019-d. Int J Dev Neurosci. 1995. PMID: 7572287 Review.
Cited by
-
Kir4.1 potassium channel subunit is crucial for oligodendrocyte development and in vivo myelination.J Neurosci. 2001 Aug 1;21(15):5429-38. doi: 10.1523/JNEUROSCI.21-15-05429.2001. J Neurosci. 2001. PMID: 11466414 Free PMC article.
-
Regulation of mitogen-activated protein kinases by sphingolipid products in oligodendrocytes.J Neurosci. 1999 Sep 1;19(17):7458-67. doi: 10.1523/JNEUROSCI.19-17-07458.1999. J Neurosci. 1999. PMID: 10460252 Free PMC article.
-
Sphingolipids in multiple sclerosis.Neuromolecular Med. 2010 Dec;12(4):351-61. doi: 10.1007/s12017-010-8128-4. Epub 2010 Jul 7. Neuromolecular Med. 2010. PMID: 20607622 Free PMC article. Review.
-
Ceramide, a putative second messenger for nerve growth factor, modulates the TTX-resistant Na(+) current and delayed rectifier K(+) current in rat sensory neurons.J Physiol. 2002 Oct 15;544(2):385-402. doi: 10.1113/jphysiol.2002.024265. J Physiol. 2002. PMID: 12381813 Free PMC article.
-
Pivotal role for acidic sphingomyelinase in cerebral ischemia-induced ceramide and cytokine production, and neuronal apoptosis.J Mol Neurosci. 2000 Oct;15(2):85-97. doi: 10.1385/JMN:15:2:85. J Mol Neurosci. 2000. PMID: 11220788
References
-
- Avruch J, Zhang X-F, Kyriakis JM. Raf meets Ras: completing the framework of a signal transduction pathway. Trends Biochem Sci. 1994;19:279–283. - PubMed
-
- Casaccia-Bonnefil P, Aibel L, Chao MV. Central glial and neuronal populations display differential sensitivity to ceramide-dependent cell death. J Neurosci Res. 1996a;43:382–389. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
