Localization of the kinesin-like protein Xklp2 to spindle poles requires a leucine zipper, a microtubule-associated protein, and dynein
- PMID: 9813089
- PMCID: PMC2148133
- DOI: 10.1083/jcb.143.3.673
Localization of the kinesin-like protein Xklp2 to spindle poles requires a leucine zipper, a microtubule-associated protein, and dynein
Abstract
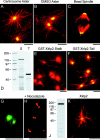
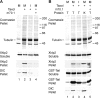
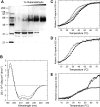
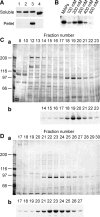
Xklp2 is a plus end-directed Xenopus kinesin-like protein localized at spindle poles and required for centrosome separation during spindle assembly in Xenopus egg extracts. A glutathione-S-transferase fusion protein containing the COOH-terminal domain of Xklp2 (GST-Xklp2-Tail) was previously found to localize to spindle poles (Boleti, H., E. Karsenti, and I. Vernos. 1996. Cell. 84:49-59). Now, we have examined the mechanism of localization of GST-Xklp2-Tail. Immunofluorescence and electron microscopy showed that Xklp2 and GST-Xklp2-Tail localize specifically to the minus ends of spindle pole and aster microtubules in mitotic, but not in interphase, Xenopus egg extracts. We found that dimerization and a COOH-terminal leucine zipper are required for this localization: a single point mutation in the leucine zipper prevented targeting. The mechanism of localization is complex and two additional factors in mitotic egg extracts are required for the targeting of GST-Xklp2-Tail to microtubule minus ends: (a) a novel 100-kD microtubule-associated protein that we named TPX2 (Targeting protein for Xklp2) that mediates the binding of GST-Xklp2-Tail to microtubules and (b) the dynein-dynactin complex that is required for the accumulation of GST-Xklp2-Tail at microtubule minus ends. We propose two molecular mechanisms that could account for the localization of Xklp2 to microtubule minus ends.
Figures





References
-
- Afshar K, Barton NR, Hawley RS, Goldstein LS. DNA binding and meiotic chromosomal localization of the Drosophilanod kinesin-like protein. Cell. 1995a;81:129–138. - PubMed
-
- Ashford, A.J., S.S.L. Andersen, and A.A. Hyman. 1998. Preparation of tubulin from bovine brain. In Cell Biology: A Laboratory Handbook. Vol. 2. J.E. Celis, editor. Academic Press, San Diego, CA. 205–212.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

