The transcription factor AP-1 is required for EGF-induced activation of rho-like GTPases, cytoskeletal rearrangements, motility, and in vitro invasion of A431 cells
- PMID: 9817764
- PMCID: PMC2132955
- DOI: 10.1083/jcb.143.4.1087
The transcription factor AP-1 is required for EGF-induced activation of rho-like GTPases, cytoskeletal rearrangements, motility, and in vitro invasion of A431 cells
Abstract
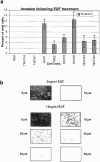
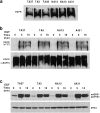
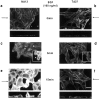
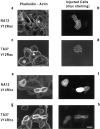
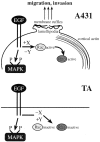
Human squamous cell carcinomas (SCC) frequently express elevated levels of epidermal growth factor receptor (EGFR). EGFR overexpression in SCC-derived cell lines correlates with their ability to invade in an in vitro invasion assay in response to EGF, whereas benign epidermal cells, which express low levels of EGFR, do not invade. EGF-induced invasion of SCC-derived A431 cells is inhibited by sustained expression of the dominant negative mutant of c-Jun, TAM67, suggesting a role for the transcription factor AP-1 (activator protein-1) in regulating invasion. Significantly, we establish that sustained TAM67 expression inhibits growth factor-induced cell motility and the reorganization of the cytoskeleton and cell-shape changes essential for this process: TAM67 expression inhibits EGF-induced membrane ruffling, lamellipodia formation, cortical actin polymerization and cell rounding. Introduction of a dominant negative mutant of Rac and of the Rho inhibitor C3 transferase into A431 cells indicates that EGF-induced membrane ruffling and lamellipodia formation are regulated by Rac, whereas EGF-induced cortical actin polymerization and cell rounding are controlled by Rho. Constitutively activated mutants of Rac or Rho introduced into A431 or A431 cells expressing TAM67 (TA cells) induce equivalent actin cytoskeletal rearrangements, suggesting that the effector pathways downstream of Rac and Rho required for these responses are unimpaired by sustained TAM67 expression. However, EGF-induced translocation of Rac to the cell membrane, which is associated with its activation, is defective in TA cells. Our data establish a novel link between AP-1 activity and EGFR activation of Rac and Rho, which in turn mediate the actin cytoskeletal rearrangements required for cell motility and invasion.
Figures








Similar articles
-
Growth factor induced activation of Rho and Rac GTPases and actin cytoskeletal reorganization in human lens epithelial cells.Mol Vis. 2003 Jul 17;9:329-36. Mol Vis. 2003. PMID: 12876554
-
Role of actin polymerization and adhesion to extracellular matrix in Rac- and Rho-induced cytoskeletal reorganization.J Cell Biol. 1997 Aug 25;138(4):913-26. doi: 10.1083/jcb.138.4.913. J Cell Biol. 1997. PMID: 9265656 Free PMC article.
-
Expression of dominant negative Jun inhibits elevated AP-1 and NF-kappaB transactivation and suppresses anchorage independent growth of HPV immortalized human keratinocytes.Oncogene. 1998 May 28;16(21):2711-21. doi: 10.1038/sj.onc.1201798. Oncogene. 1998. PMID: 9652737
-
Ras-related GTPases and the cytoskeleton.Mol Biol Cell. 1992 May;3(5):475-9. doi: 10.1091/mbc.3.5.475. Mol Biol Cell. 1992. PMID: 1611153 Free PMC article. Review.
-
Signal transduction through the GTP-binding proteins Rac and Rho.J Cell Sci Suppl. 1994;18:127-31. doi: 10.1242/jcs.1994.supplement_18.19. J Cell Sci Suppl. 1994. PMID: 7883787 Review.
Cited by
-
Phosphoinositide 3-Kinase C2beta regulates cytoskeletal organization and cell migration via Rac-dependent mechanisms.Mol Biol Cell. 2006 Sep;17(9):3729-44. doi: 10.1091/mbc.e05-11-1083. Epub 2006 Jun 14. Mol Biol Cell. 2006. PMID: 16775008 Free PMC article.
-
The role of AP-1 in self-sufficient proliferation and migration of cancer cells and its potential impact on an autocrine/paracrine loop.Oncotarget. 2018 Sep 28;9(76):34259-34278. doi: 10.18632/oncotarget.26047. eCollection 2018 Sep 28. Oncotarget. 2018. PMID: 30344941 Free PMC article.
-
Critical role of c-Jun overexpression in liver metastasis of human breast cancer xenograft model.BMC Cancer. 2007 Aug 1;7:145. doi: 10.1186/1471-2407-7-145. BMC Cancer. 2007. PMID: 17672916 Free PMC article.
-
Apoptosis, growth arrest and suppression of invasiveness by CRE-decoy oligonucleotide in ovarian cancer cells: protein kinase A downregulation and cytoplasmic export of CRE-binding proteins.Mol Cell Biochem. 2001 Feb;218(1-2):55-63. doi: 10.1023/a:1007205205131. Mol Cell Biochem. 2001. PMID: 11330838
-
Transducer of Cdc42-dependent actin assembly promotes epidermal growth factor-induced cell motility and invasiveness.J Biol Chem. 2011 Jan 21;286(3):2261-72. doi: 10.1074/jbc.M110.157974. Epub 2010 Nov 9. J Biol Chem. 2011. PMID: 21062739 Free PMC article.
References
-
- Anand-Apte B, Zetter BR, Viswanathan A, Qiu RG, Chen J, Ruggieri R, Symons M. Platelet-derived growth factor and fibronectin-stimulated migration are differentially regulated by the Rac and extracellular signal-regulated kinase pathways. J Biol Chem. 1997;272:30688–30692. - PubMed
-
- Birchmeier W. E-cadherin as a tumor (invasion) suppressor gene. Bioessays. 1995;17:97–99. - PubMed
-
- Bretscher A, Reczek D, Berryman M. Ezrin: a protein requiring conformational activation to link microfilaments to the plasma membrane in the assembly of cell surface structures. J Cell Sci. 1997;110:3011–3018. - PubMed
-
- Brown PH, Alani R, Preis LH, Szabo E, Birrer MJ. Suppression of oncogene-induced transformation by a deletion mutant of c-jun. Oncogene. 1993;8:877–886. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

