Stability of the Agrobacterium tumefaciens VirB10 protein is modulated by growth temperature and periplasmic osmoadaption
- PMID: 9852004
- PMCID: PMC107763
- DOI: 10.1128/JB.180.24.6597-6606.1998
Stability of the Agrobacterium tumefaciens VirB10 protein is modulated by growth temperature and periplasmic osmoadaption
Abstract
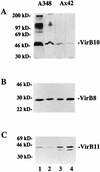
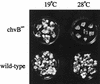
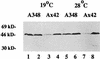
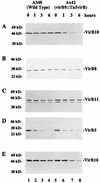
Export of oncogenic T-DNA from the phytopathogen Agrobacterium tumefaciens is mediated by the products of the virB operon. It has recently been reported (K. J. Fullner and E. W. Nester, J. Bacteriol. 178:1498-1504, 1996) that DNA transfer does not occur at elevated temperatures; these observations correlate well with much earlier studies on the temperature sensitivity of crown gall tumor development on plants. In testing the hypothesis that this loss of DNA movement reflects a defect in assembly or maintenance of a stable DNA transfer machinery at high temperature, we have found that steady-state levels of VirB10 are sensitive to growth temperature while levels of several other VirB proteins are considerably less affected. This temperature-dependent failure to accumulate VirB10 is exacerbated in an attachment-deficient mutant strain (chvB) which exhibits pleiotropic defects in periplasmic osmoadaption, and virulence of a chvB mutant can be partially restored by lowering the temperature at which the bacteria and the plant tissue are cocultivated. Furthermore, the stability of VirB10 is diminished in cells lacking functional VirB9, but only under conditions of low osmolarity. We propose that newly synthesized VirB10 is inherently labile in the presence of a large osmotic gradient across the inner membrane and is rapidly degraded unless it is stabilized by VirB9-dependent assembly into oligomeric complexes. The possibility that VirB10-containing complexes are not assembled properly at elevated temperatures suggests an explanation for the decades-old observation that tumor formation is exquisitely sensitive to ambient temperature.
Figures



References
-
- Ankenbauer R G, Best E A, Palanca C A, Nester E W. Mutants of the Agrobacterium tumefaciens virA gene exhibiting acetosyringone-independent expression of the vir regulon. Mol Plant-Microbe Interact. 1991;4:400–406. - PubMed
-
- Banta, L. M. Unpublished data.
-
- Baron C, Zambryski P C. The plant response in pathogenesis, symbiosis, and wounding: variations on a common theme? Annu Rev Genet. 1995;29:107–129. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

