pEg7, a new Xenopus protein required for mitotic chromosome condensation in egg extracts
- PMID: 9852142
- PMCID: PMC2132990
- DOI: 10.1083/jcb.143.6.1437
pEg7, a new Xenopus protein required for mitotic chromosome condensation in egg extracts
Abstract
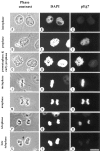
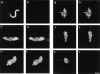
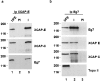
We have isolated a cDNA, Eg7, corresponding to a Xenopus maternal mRNA, which is polyadenylated in mature oocytes and deadenylated in early embryos. This maternal mRNA encodes a protein, pEg7, whose expression is strongly increased during oocyte maturation. The tissue and cell expression pattern of pEg7 indicates that this protein is only readily detected in cultured cells and germ cells. Immunolocalization in Xenopus cultured cells indicates that pEg7 concentrates onto chromosomes during mitosis. A similar localization of pEg7 is observed when sperm chromatin is allowed to form mitotic chromosomes in cytostatic factor-arrested egg extracts. Incubating these extracts with antibodies directed against two distinct parts of pEg7 provokes a strong inhibition of the condensation and resolution of mitotic chromosomes. Biochemical experiments show that pEg7 associates with Xenopus chromosome-associated polypeptides C and E, two components of the 13S condensin.
Figures




References
-
- Adachi Y, Luke M, Laemmli UK. Chromosome assembly in vitro: topoisomerase II is required for condensation. Cell. 1991;64:137–148. - PubMed
-
- Bhat MA, Philp AV, Glover DM, Bellen HJ. Chromatid segregation at anaphase requires the barren product, a novel chromosome-associated protein that interacts with Topoisomerase II. Cell. 1996;87:1103–1114. - PubMed
-
- Bravo R, Knowland J. Classes of proteins synthesized in oocytes, eggs, embryos, and differentiated tissues of Xenopus laevis. . Differentiation. 1979;13:101–108. - PubMed
-
- Gasser SM. Chromosome structure. Coiling up chromosomes. Curr Biol. 1995;5:357–360. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

