Two functional states of the CD11b A-domain: correlations with key features of two Mn2+-complexed crystal structures
- PMID: 9852148
- PMCID: PMC2132978
- DOI: 10.1083/jcb.143.6.1523
Two functional states of the CD11b A-domain: correlations with key features of two Mn2+-complexed crystal structures
Abstract
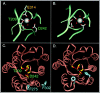
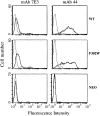
In the presence of bound Mn2+, the three- dimensional structure of the ligand-binding A-domain from the integrin CR3 (CD11b/CD18) is shown to exist in the "open" conformation previously described only for a crystalline Mg2+ complex. The open conformation is distinguished from the "closed" form by the solvent exposure of F302, a direct T209-Mn2+ bond, and the presence of a glutamate side chain in the MIDAS site. Approximately 10% of wild-type CD11b A-domain is present in an "active" state (binds to activation-dependent ligands, e.g., iC3b and the mAb 7E3). In the isolated domain and in the holoreceptor, the percentage of the active form can be quantitatively increased or abolished in F302W and T209A mutants, respectively. The iC3b-binding site is located on the MIDAS face and includes conformationally sensitive residues that undergo significant shifts in the open versus closed structures. We suggest that stabilization of the open structure is independent of the nature of the metal ligand and that the open conformation may represent the physiologically active form.
Figures



References
-
- Altieri DC. Occupancy of CD11b/CD18 (Mac-1) divalent ion binding site(s) induces leukocyte adhesion. J Immunol. 1991;147:1891–1896. - PubMed
-
- Altieri DC, Edgington TS. The saturable high affinity association of factor X to ADP-stimulated monocytes defines a novel function of the MAC-1 receptor. J Biol Chem. 1988;263:7007–7015. - PubMed
-
- Andrew DP, Berlin C, Honda S, Yoshino T, Hamann A, Holzmann B, Kilshaw PJ, Butcher EC. Distinct but overlapping epitopes are involved in alpha 4 beta 7-mediated adhesion to vascular cell adhesion molecule-1, mucosal addressin-1, fibronectin, and lymphocyte aggregation. J Immunol. 1994;153:3847–3861. - PubMed
-
- Bajt ML, Loftus JC. Mutation of a ligand binding domain of β3 integrin: integral role of oxygenated residues in αIIbβ3 (GPIIb-IIIa) receptor function. J Biol Chem. 1994;269:20913–20919. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

