Calcium currents of rhythmic neurons recorded in the isolated respiratory network of neonatal mice
- PMID: 9852600
- PMCID: PMC6793347
- DOI: 10.1523/JNEUROSCI.18-24-10652.1998
Calcium currents of rhythmic neurons recorded in the isolated respiratory network of neonatal mice
Abstract
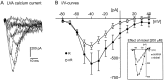
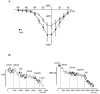
To obtain a quantitative characterization of voltage-activated calcium currents in respiratory neurons, we performed voltage-clamp recordings in the transverse brainstem slice of mice from neurons located within the ventral respiratory group. It is assumed that this medullary region contains the neuronal network responsible for generating the respiratory rhythm. This study represents one of the first attempts to analyze quantitatively the currents in respiratory neurons. The inward calcium currents of VRG neurons consisted of two components: a high voltage-activated (HVA) and a low voltage-activated (LVA) calcium current. The activation threshold of the HVA current was at -40 mV. It was fully activated (peak voltage) between -10 and 0 mV. The half-maximal activation (V50) was at -27. 29 mV +/- 1.15 (n = 24). The HVA current was inactivated completely at a holding potential of -35 mV and fully deinactivated at a holding potential of -65 mV (V50, -52.26 mV +/- 0.27; n = 18). The threshold for the activation of the LVA current was at -65 mV. This current had its peak voltage between -50 and -40 mV (mean, V50 = -59. 15 mV +/- 0.21; n = 15). The LVA current was inactivated completely at a holding potential of -65 mV and deinactivated fully at a holding potential of -95 mV (mean, V50 = -82.40 mV +/- 0.32; n = 38). These properties are consistent with other studies suggesting that the LVA current is a T-type current. The properties of these inward currents are discussed with respect to their role in generating Ca2+ potentials that may contribute to the generation of the mammalian respiratory rhythm.
Figures








Comment on
-
Fundic gland polyps under omeprazole treatment.Am J Clin Pathol. 1999 Oct;112(4):576-7. doi: 10.1093/ajcp/112.4.576. Am J Clin Pathol. 1999. PMID: 10510676 No abstract available.
References
-
- Armstrong CM, Gilly WF. Access resistance and space clamp problems associated with whole-cell patch clamping. Methods Enzymol. 1992;207:100–122. - PubMed
-
- Bean BP. Classes of calcium channels in vertebrate cells. Annu Rev Physiol. 1989;51:367–384. - PubMed
-
- Beck H, Steffens R, Heinemann U, Elger CE. Properties of voltage-activated Ca2+ currents in acutely isolated human hippocampal granule cells. J Neurophysiol. 1997;77:1526–1537. - PubMed
-
- Bianchi AL, Denavit-Saubie M, Champagnat J. Central control of breathing in mammals: neuronal circuitry, membrane properties, and neurotransmitters. Physiol Rev. 1995;75:1–45. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
