Variables that affect assays for plasma cytokines and soluble activation markers
- PMID: 9874670
- PMCID: PMC95666
- DOI: 10.1128/CDLI.6.1.89-95.1999
Variables that affect assays for plasma cytokines and soluble activation markers
Abstract
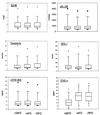
Cytokines and soluble immune activation markers that reflect cytokine activities in vivo are increasingly being measured in plasma, serum, and other body fluids. They provide useful diagnostic and prognostic information as well as insight into disease pathogenesis. Assays of neopterin, beta2-microglobulin, soluble interleukin-2 receptor, and soluble tumor necrosis factor receptor type II as well as of the cytokines tumor necrosis factor alpha and gamma interferon (IFN-gamma) were evaluated by using serum and plasma samples of human immunodeficiency virus (HIV)-positive and HIV-negative subjects. Many factors were found to influence the outcomes of these assays. Substantial differences in apparent levels of analytes were frequently found when enzyme-linked immunosorbent assay (ELISA) kits from different manufacturers were used. In some cases, differences were found in the standards provided by separate manufacturers. Furthermore, the analytic results from different lots of ELISA kits supplied by single manufacturers differed by as much as 50%. The need for uniformity in the standards for quantitative assays was clearly illustrated. International reference standards are available for cytokines but not for soluble cytokine receptors or soluble activation markers. Marker levels in serum or in plasma were similar except those for IFN-gamma. Most of the analytes were stable under several storage conditions. Thus, batch testing of frozen stored samples is feasible. The findings indicate that for longitudinal studies, the levels of cytokines and immune activation markers in plasma or serum should be measured by using preverified reagents from one manufacturer. The quality of laboratory performance can have an impact on clinical relevance. Proficiency testing and external quality assurance programs can help to develop the needed consensus.
Figures

References
-
- Aggarwal B B, Puri R K. Common and uncommon features of cytokines and cytokine receptors: an overview. In: Aggarwal B B, Puri R K, editors. Human cytokines: their role in disease and therapy. Cambridge, Mass: Blackwell Scientific; 1995. pp. 3–24.
-
- Aukrust P, Liabakk N B, Müller F, Lien E, Espevik T, Froland S S. Serum levels of tumor necrosis factor-alpha (TNF-α) and soluble TNF receptors in human immunodeficiency virus type 1 infection: correlations to clinical immunologic and virologic parameters. J Infect Dis. 1994;169:420–424. - PubMed
-
- Bienvenu J, Coulon L, Doche C, Gutowski M C, Grau G. Analytical performances of commercial ELISA kits for IL-2, IL-6 and TNFα. A WHO study. Eur Cytokine Netw. 1993;4:447–451. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

