A small number of residues in the class II molecule I-Au confer the ability to bind the myelin basic protein peptide Ac1-11
- PMID: 9874795
- PMCID: PMC15116
- DOI: 10.1073/pnas.96.1.197
A small number of residues in the class II molecule I-Au confer the ability to bind the myelin basic protein peptide Ac1-11
Abstract
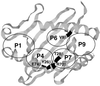
The N-terminal peptide Ac1-11 of myelin basic protein induces experimental autoimmune encephalomyelitis in H-2(u) and (H-2(u) x H-2(s)) mice but does not in H-2(s) mice. Ac1-11 binds weakly to the class II major histocompatibility complex (MHC) molecule I-Au but not at all to I-As. We have studied the interaction of Ac1-11 and I-Au as a model system for therapeutic intervention in the autoimmune response seen in experimental autoimmune encephalomyelitis. Two polymorphic residues that differ between I-Au and I-As, Y26beta and T28beta, and one conserved residue, E74beta, confer specific binding of Ac1-11 to I-Au. A fourth residue, R70beta in I-Au, affects both peptide binding and T cell recognition. These results are consistent with a model that places arginine at position five of Ac1-11 in pockets 4 and 7 of the MHC groove, which is formed in part by residues 26, 28, 70, and 74 of Abetau and places lysine at position four of Ac1-11, previously shown to be a major MHC contact, in hydrophobic pocket 6. The data indicate that the primary region of I-Au that confers specific binding of Ac1-11 lies in the center of the peptide binding groove rather than in the region that contacts the N terminus of the peptide, as has been shown for HLA DR and the homologous I-E molecules.
Figures
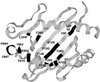

Similar articles
-
T cell recognition of distinct peptide:I-Au conformers in murine experimental autoimmune encephalomyelitis.J Immunol. 2003 Sep 1;171(5):2467-77. doi: 10.4049/jimmunol.171.5.2467. J Immunol. 2003. PMID: 12928395
-
Evidence that the autoimmune antigen myelin basic protein (MBP) Ac1-9 binds towards one end of the major histocompatibility complex (MHC) cleft.J Exp Med. 1998 May 4;187(9):1505-16. doi: 10.1084/jem.187.9.1505. J Exp Med. 1998. PMID: 9565642 Free PMC article.
-
Interactions among polymorphic and conserved residues in MHC class II proteins affect MHC-peptide conformation and T cell recognition.Int Immunol. 1995 May;7(5):747-61. doi: 10.1093/intimm/7.5.747. Int Immunol. 1995. PMID: 7547702
-
MHC class II peptide flanking residues of exogenous antigens influence recognition by autoreactive T cells.Autoimmun Rev. 2003 Jan;2(1):8-12. doi: 10.1016/s1568-9972(02)00102-7. Autoimmun Rev. 2003. PMID: 12848969 Review.
-
T cell recognition in experimental autoimmune encephalomyelitis: prospects for immune intervention with synthetic peptides.Int Rev Immunol. 1990;6(1):37-47. doi: 10.3109/08830189009056616. Int Rev Immunol. 1990. PMID: 1715375 Review.
Cited by
-
CD4+CD25+ regulatory T cells limit the risk of autoimmune disease arising from T cell receptor crossreactivity.Proc Natl Acad Sci U S A. 2005 Nov 29;102(48):17418-23. doi: 10.1073/pnas.0507454102. Epub 2005 Nov 15. Proc Natl Acad Sci U S A. 2005. PMID: 16287973 Free PMC article.
-
Negative selection during the peripheral immune response to antigen.J Exp Med. 2001 Jan 1;193(1):1-11. doi: 10.1084/jem.193.1.1. J Exp Med. 2001. PMID: 11136816 Free PMC article.
-
Peptide-based immunotherapy of autoimmunity: a path of puzzles, paradoxes and possibilities.Immunology. 2001 Dec;104(4):367-76. doi: 10.1046/j.1365-2567.2001.01324.x. Immunology. 2001. PMID: 11899421 Free PMC article. Review. No abstract available.
-
A previously unappreciated polymorphism in the beta chain of I-As expressed in autoimmunity-prone SJL mice: Combined impact on antibody, CD4 T cell recognition and MHC class II dimer structural stability.Mol Immunol. 2022 Mar;143:17-26. doi: 10.1016/j.molimm.2021.12.022. Epub 2022 Jan 5. Mol Immunol. 2022. PMID: 34995990 Free PMC article.
References
-
- Zamvil S, Steinman L. Annu Rev Immunol. 1990;8:579–621. - PubMed
-
- Sobel R A, Tuohy V K, Lu Z J, Laursen R A, Lees M B. J Neuropathol Exp Neurol. 1990;49:468–479. - PubMed
-
- Wraith D C, Smilek D E, Mitchell D J, Steinman L, McDevitt H O. Cell. 1989;59:247–255. - PubMed
-
- Gautam A M, Pearson C I, Sinha A A, Smilek D E, Steinman L, McDevitt H O. J Immunol. 1992;148:3049–3054. - PubMed
-
- Mason K, Denney D W, Jr, McConnell H M. J Immunol. 1995;154:5216–5227. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

