Limited protection from a pathogenic chimeric simian-human immunodeficiency virus challenge following immunization with attenuated simian immunodeficiency virus
- PMID: 9882330
- PMCID: PMC103949
- DOI: 10.1128/JVI.73.2.1262-1270.1999
Limited protection from a pathogenic chimeric simian-human immunodeficiency virus challenge following immunization with attenuated simian immunodeficiency virus
Abstract
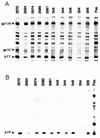
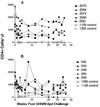
Two live attenuated single-deletion mutant simian immunodeficiency virus (SIV) constructs, SIV239Deltanef and SIVPBj6.6Deltanef, were tested for their abilities to stimulate protective immunity in macaques. During the immunization period the animals were examined for specific immune responses and virus growth. Each construct generated high levels of specific immunity in all of the immunized animals. The SIV239Deltanef construct was found to grow to high levels in all immunized animals, with some animals remaining positive for virus isolation and plasma RNA throughout the immunization period. The SIVPBj6.6Deltanef was effectively controlled by all of the immunized animals, with virus mostly isolated only during the first few months following immunization and plasma RNA never detected. Following an extended period of immunization of over 80 weeks, the animals were challenged with a pathogenic simian-human immunodeficiency virus (SHIV) isolate, SIV89. 6PD, by intravenous injection. All of the SIV239Deltanef-immunized animals became infected with the SHIV isolate; two of five animals eventually controlled the challenge and three of five animals, which failed to check the immunizing virus, progressed to disease state before the unvaccinated controls. One of five animals immunized with SIVPBj6.6Deltanef totally resisted infection by the challenge virus, while three others limited its growth and the remaining animal became persistently infected and eventually died of a pulmonary thrombus. These data indicate that vaccination with attenuated SIV can protect macaques from disease and in some cases from infection by a divergent SHIV. However, if animals are unable to control the immunizing virus, potential damage that can accelerate the disease course of a pathogenic challenge virus may occur.
Figures



References
-
- Agy M B, Frumkin L R, Corey L, Coombs R W, Wolinsky S M, Koehler J, Morton W R, Katze M G. Infection of Macaca nemestrina by human immunodeficiency virus type-1. Science. 1992;257:103–106. - PubMed
-
- Almond N, Kent K, Cranage M, Rud E, Clarke B, Stott E J. Protection by attenuated simian immunodeficiency virus in macaques against challenge with virus-infected cells. Lancet. 1995;345:1342–1344. - PubMed
-
- Almond N, Rose J, Sangster R, Silvera P, Stebbings R, Walker B, Stott E J. Mechanisms of protection induced by attenuated simian immunodeficiency virus. 1. Protection cannot be transferred with immune serum. J Gen Virol. 1997;78:1919–1922. - PubMed
-
- Baba T W, Jeong Y S, Penninck D, Bronson R, Greene M F, Ruprecht R M. Pathogenicity of live, attenuated SIV after mucosal infection of neonatal macaques. Science. 1995;267:1820–1825. - PubMed
-
- Benveniste R E, Roodman S T, Hill R W, Knott W B, Ribas J L, Lewis M G, Eddy G A. Infectivity of titered doses of simian immunodeficiency virus clone E11S inoculated intravenously into rhesus macaques (Macaca mulatta) J Med Primatol. 1994;23:83–88. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

