Regulatory T cells in the control of autoimmunity: the essential role of transforming growth factor beta and interleukin 4 in the prevention of autoimmune thyroiditis in rats by peripheral CD4(+)CD45RC- cells and CD4(+)CD8(-) thymocytes
- PMID: 9892610
- PMCID: PMC2192980
- DOI: 10.1084/jem.189.2.279
Regulatory T cells in the control of autoimmunity: the essential role of transforming growth factor beta and interleukin 4 in the prevention of autoimmune thyroiditis in rats by peripheral CD4(+)CD45RC- cells and CD4(+)CD8(-) thymocytes
Abstract
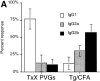
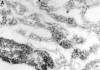
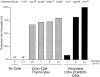
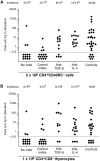
Previous studies have shown that induction of autoimmune diabetes by adult thymectomy and split dose irradiation of PVG.RT1(u) rats can be prevented by their reconstitution with peripheral CD4(+)CD45RC-TCR-alpha/beta+RT6(+) cells and CD4(+)CD8(-) thymocytes from normal syngeneic donors. These data provide evidence for the role of regulatory T cells in the prevention of a tissue-specific autoimmune disease but the mode of action of these cells has not been reported previously. In this study, autoimmune thyroiditis was induced in PVG.RT1(c) rats using a similar protocol of thymectomy and irradiation. Although a cell-mediated mechanism has been implicated in the pathogenesis of diabetes in PVG.RT1(u) rats, development of thyroiditis is independent of CD8(+) T cells and is characterized by high titers of immunoglobulin (Ig)G1 antithyroglobulin antibodies, indicating a major humoral component in the pathogenesis of disease. As with autoimmune diabetes in PVG. RT1(u) rats, development of thyroiditis was prevented by the transfer of CD4(+)CD45RC- and CD4(+)CD8(-) thymocytes from normal donors but not by CD4(+)CD45RC+ peripheral T cells. We now show that transforming growth factor (TGF)-beta and interleukin (IL)-4 both play essential roles in the mechanism of this protection since administration of monoclonal antibodies that block the biological activity of either of these cytokines abrogates the protective effect of the donor cells in the recipient rats. The prevention of both diabetes and thyroiditis by CD4(+)CD45RC- peripheral cells and CD4(+)CD8(-) thymocytes therefore does not support the view that the mechanism of regulation involves a switch from a T helper cell type 1 (Th1) to a Th2-like response, but rather relies upon a specific suppression of the autoimmune responses involving TGF-beta and IL-4. The observation that the same two cytokines were implicated in the protective mechanism, whether thymocytes or peripheral cells were used to prevent autoimmunity, strongly suggests that the regulatory cells from both sources act in the same way and that the thymocytes are programmed in the periphery for their protective role. The implications of this result with respect to immunological homeostasis are discussed.
Figures



Similar articles
-
Peripheral autoantigen induces regulatory T cells that prevent autoimmunity.J Exp Med. 1999 Mar 1;189(5):877-82. doi: 10.1084/jem.189.5.877. J Exp Med. 1999. PMID: 10049952 Free PMC article.
-
CD4+CD8- thymocytes that express L-selectin protect rats from diabetes upon adoptive transfer.Eur J Immunol. 1996 Nov;26(11):2702-8. doi: 10.1002/eji.1830261123. Eur J Immunol. 1996. PMID: 8921958
-
Infiltrating CD45RC- T cells are associated with immunologic unresponsiveness induced by donor class I major histocompatibility complex antigens in rats.Hepatology. 1998 Aug;28(2):450-8. doi: 10.1002/hep.510280224. Hepatology. 1998. PMID: 9696011
-
The role of subsets of CD4+ T cells in autoimmunity.Ciba Found Symp. 1995;195:173-82; discussion 182-8. doi: 10.1002/9780470514849.ch12. Ciba Found Symp. 1995. PMID: 8724837 Review.
-
Aetiopathogenesis of autoimmune hepatitis.World J Gastroenterol. 2008 Jun 7;14(21):3306-12. doi: 10.3748/wjg.14.3306. World J Gastroenterol. 2008. PMID: 18528928 Free PMC article. Review.
Cited by
-
Activated CD4+CD25+ T cells selectively kill B lymphocytes.Blood. 2006 May 15;107(10):3925-32. doi: 10.1182/blood-2005-11-4502. Epub 2006 Jan 17. Blood. 2006. PMID: 16418326 Free PMC article.
-
Allograft rejection results from a failed attempt by the immune system to protect foreign tissue.Immunol Res. 2000;21(2-3):177-83. doi: 10.1385/ir:21:2-3:177. Immunol Res. 2000. PMID: 10852115 Review.
-
Dysfunctions, Molecular Mechanisms, and Therapeutic Strategies of Regulatory T Cells in Rheumatoid Arthritis.Front Pharmacol. 2021 Aug 26;12:716081. doi: 10.3389/fphar.2021.716081. eCollection 2021. Front Pharmacol. 2021. PMID: 34512345 Free PMC article. Review.
-
In vivo dynamics of antigen-specific regulatory T cells not predicted from behavior in vitro.Proc Natl Acad Sci U S A. 2003 Jul 22;100(15):8886-91. doi: 10.1073/pnas.1533365100. Epub 2003 Jul 11. Proc Natl Acad Sci U S A. 2003. PMID: 12857946 Free PMC article.
-
Tumor-specific immunotherapy targeting the EGFRvIII mutation in patients with malignant glioma.Semin Immunol. 2008 Oct;20(5):267-75. doi: 10.1016/j.smim.2008.04.001. Epub 2008 Jun 9. Semin Immunol. 2008. PMID: 18539480 Free PMC article. Review.
References
-
- Taguchi O, Takahashi T. Administration of anti-interleukin-2 receptor alpha antibody in vivo induces localized autoimmune disease. Eur J Immunol. 1996;26:1608–1612. - PubMed
-
- Bonomo A, Kehn P, Shevach E. Post-thymectomy autoimmunity: abnormal T-cell homeostasis. Immunol Today. 1995;16:61–67. - PubMed
-
- Saoudi A, Seddon B, Heath V, Fowell V, Mason D. The physiological role of regulatory T cells in the prevention of autoimmunity: the function of the thymus in the generation of the regulatory T cell subset. Immunol Rev. 1996;149:195–216. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

