Role of decay-accelerating factor in regulating complement activation on the erythrocyte surface as revealed by gene targeting
- PMID: 9892684
- PMCID: PMC15187
- DOI: 10.1073/pnas.96.2.628
Role of decay-accelerating factor in regulating complement activation on the erythrocyte surface as revealed by gene targeting
Abstract
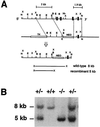
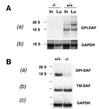
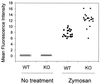
Decay-accelerating factor (DAF) is a glycosylphosphatidylinositol (GPI)-anchored membrane protein that inhibits both the classical and the alternative pathways of complement activation. DAF has been studied extensively in humans under two clinical settings: when absent from the erythrocytes of paroxysmal nocturnal hemoglobinuria (PNH) patients, who suffer from complement-mediated hemolytic anemia, and in transgenic pigs expressing human DAF, which have been developed to help overcome complement-mediated hyperacute rejection in xenotransplantation. Nevertheless, the exact role of DAF in regulating complement activation in vivo on the cell surface and the species specificity of this molecule remain to be fully characterized. To address these issues, we have used gene targeting to produce mice lacking GPI-anchored DAF. We found that erythrocytes from mice deficient in GPI-anchored DAF showed no increase in spontaneous complement activation in vivo but exhibited impaired regulation of zymosan-initiated bystander and antibody-triggered classical pathway complement activation in vitro, resulting in enhanced complement deposition. Despite a high level of C3 fixation, no homologous hemolysis occurred. It is noteworthy that GPI-linked DAF knockout erythrocytes, when tested with human and guinea pig sera, were more susceptible to heterologous complement lysis than were normal erythrocytes. These results suggest that DAF is capable of regulating homologous as well as heterologous complement activation via the alternative or the classical pathway. They also indicate that DAF deficiency alone is not sufficient to cause homologous hemolysis. In contrast, when the assembly of the membrane-attack complex is not properly regulated, as in the case of heterologous complement activation or in PNH patients, impaired erythrocyte DAF activity and enhanced C3 deposition could lead to increased hemolytic reaction.
Figures

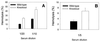
Similar articles
-
Crry, but not CD59 and DAF, is indispensable for murine erythrocyte protection in vivo from spontaneous complement attack.Blood. 2002 May 15;99(10):3707-16. doi: 10.1182/blood.v99.10.3707. Blood. 2002. PMID: 11986227
-
Molecular basis of the enhanced susceptibility of the erythrocytes of paroxysmal nocturnal hemoglobinuria to hemolysis in acidified serum.Blood. 1991 Aug 1;78(3):820-9. Blood. 1991. PMID: 1713516
-
Test for ability of decay-accelerating factor (DAF, CD55) and CD59 to alleviate complement-mediated damage of xeno-erythrocytes.Scand J Immunol. 1993 Jul;38(1):37-44. doi: 10.1111/j.1365-3083.1993.tb01691.x. Scand J Immunol. 1993. PMID: 7687071
-
Glycosyl phosphatidylinositol-linked blood group antigens and paroxysmal nocturnal hemoglobinuria.Transfus Clin Biol. 1995;2(4):277-90. doi: 10.1016/s1246-7820(05)80094-1. Transfus Clin Biol. 1995. PMID: 8542026 Review.
-
Congenital Defects in the Expression of the Glycosylphosphatidylinositol-Anchored Complement Regulatory Proteins CD59 and Decay-Accelerating Factor.Semin Hematol. 2018 Jul;55(3):136-140. doi: 10.1053/j.seminhematol.2018.04.004. Epub 2018 Apr 16. Semin Hematol. 2018. PMID: 30032750 Review.
Cited by
-
Expression dynamics of Crry at the implantation sites in normal pregnancy and response against miscarriage induction.J Vet Med Sci. 2023 Jan 20;85(1):92-98. doi: 10.1292/jvms.22-0286. Epub 2022 Nov 29. J Vet Med Sci. 2023. PMID: 36450590 Free PMC article.
-
Deletion of decay-accelerating factor (CD55) exacerbates autoimmune disease development in MRL/lpr mice.Am J Pathol. 2002 Sep;161(3):1077-86. doi: 10.1016/S0002-9440(10)64268-X. Am J Pathol. 2002. PMID: 12213736 Free PMC article.
-
Associations of dicarbonyl stress with complement activation: the CODAM study.Diabetologia. 2020 May;63(5):1032-1042. doi: 10.1007/s00125-020-05098-4. Epub 2020 Jan 28. Diabetologia. 2020. PMID: 31993713 Free PMC article.
-
Decay-accelerating factor induction by tumour necrosis factor-alpha, through a phosphatidylinositol-3 kinase and protein kinase C-dependent pathway, protects murine vascular endothelial cells against complement deposition.Immunology. 2003 Oct;110(2):258-68. doi: 10.1046/j.1365-2567.2003.01733.x. Immunology. 2003. PMID: 14511240 Free PMC article.
-
Role of complement and complement regulatory proteins in the complications of diabetes.Endocr Rev. 2015 Jun;36(3):272-88. doi: 10.1210/er.2014-1099. Epub 2015 Apr 10. Endocr Rev. 2015. PMID: 25859860 Free PMC article. Review.
References
-
- Volanakis J E, Frank M. The Human Complement System in Health and Disease. New York: Dekker; 1998.
-
- Lambris J D. The Third Component of Complement: Chemistry and Biology. Berlin: Springer; 1990. - PubMed
-
- Nicholson-Weller A, Wang C E. J Lab Clin Med. 1994;123:485–491. - PubMed
-
- Nicholson-Weller A, Burge J, Fearon D T, Weller P F, Austen K F. J Immunol. 1982;129:184–189. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

