Unusual carotenoid composition and a new type of xanthophyll cycle in plants
- PMID: 9927706
- PMCID: PMC15363
- DOI: 10.1073/pnas.96.3.1135
Unusual carotenoid composition and a new type of xanthophyll cycle in plants
Abstract
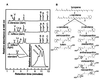
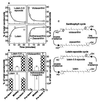
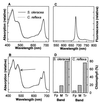
The capture of photons by the photosynthetic apparatus is the first step in photosynthesis in all autotrophic higher plants. This light capture is dominated by pigment-containing proteins known as light-harvesting complexes (LHCs). The xanthophyll-carotenoid complement of these LHCs (neoxanthin, violaxanthin, and lutein) is highly conserved, with no deletions and few, uncommon additions. We report that neoxanthin, considered an integral component of LHCs, is stoichiometrically replaced by lutein-5,6-epoxide in the parasitic angiosperm Cuscuta reflexa, without compromising the structural integrity of the LHCs. Lutein-5,6-epoxide differs from neoxanthin in that it is involved in a light-driven deepoxidation cycle similar to the deepoxidation of violaxanthin in the xanthophyll cycle, which is implicated in protection against photodamage. The absence of neoxanthin and its replacement by lutein-5,6-epoxide changes our understanding of the structure-function relationship in LHCs, has implications for biosynthetic pathways involving neoxanthin (such as the plant hormone abscisic acid), and identifies one of the early steps associated with the evolution of heterotrophy from autotrophy in plants.
Figures

References
-
- Johnson G N, Young A J, Scholes J D, Horton P. Plant Cell Environ. 1993;16:673–679.
-
- Bungard R A, McNeil D, Morton J D. Aust J Plant Physiol. 1997;24:205–214.
-
- Kühlbrandt W, Wang D N, Fujiyoshi Y. Nature (London) 1994;367:614–621. - PubMed
-
- Kühlbrandt W, Wang D N. Nature (London) 1991;350:130–134. - PubMed
-
- Lee H-S, Milborrow B V. Aust J Plant Pysiol. 1997;24:715–726.
LinkOut - more resources
Full Text Sources
Other Literature Sources

