Anti-inflammatory effects of a new tumour necrosis factor-alpha (TNF-alpha) inhibitor (CNI-1493) in collagen-induced arthritis (CIA) in rats
- PMID: 9933418
- PMCID: PMC1905187
- DOI: 10.1046/j.1365-2249.1999.00750.x
Anti-inflammatory effects of a new tumour necrosis factor-alpha (TNF-alpha) inhibitor (CNI-1493) in collagen-induced arthritis (CIA) in rats
Abstract
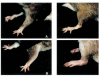
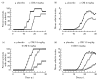
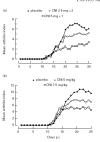
A recently developed compound, a multivalent guanylhydrazone (CNI-1493) that inhibits TNF-alpha production by suppressing TNF-alpha translational efficiency, was administered in an experimental model of collagen type II-induced arthritis in DA rats. CNI-1493 was injected daily intraperitoneally either before the onset of arthritis or after the establishment of clinical disease. Prophylactic treatment with CNI-1493 significantly prevented or delayed the onset and suppressed the severity of arthritis in a dose-dependent manner. Therapeutic intervention with CNI-1493 in established joint disease also resulted in a significant reduction of clinical signs of arthritis in treated animals. No severe side-effects were noted when animals were treated with daily CNI-1493 doses up to 5 mg/kg. An immunohistochemical study was performed which demonstrated that CNI-1493 led to a reduced expression of TNF-alpha at the site of disease activity. Thus, CNI-1493 with documented inhibitory effects on TNF-alpha synthesis, has proven successful in ameliorating the course of arthritis in CIA. We believe that the use of a compound such as CNI-1493 with a defined mode of action provides a useful tool for dissecting and understanding important pathogenic mechanisms operating in the development of chronic arthritis.
Figures

Similar articles
-
Dynamics of early synovial cytokine expression in rodent collagen-induced arthritis : a therapeutic study using a macrophage-deactivating compound.Am J Pathol. 2001 Feb;158(2):491-500. doi: 10.1016/S0002-9440(10)63991-0. Am J Pathol. 2001. PMID: 11159186 Free PMC article.
-
Protection against cartilage and bone destruction by systemic interleukin-4 treatment in established murine type II collagen-induced arthritis.Arthritis Res. 1999;1(1):81-91. doi: 10.1186/ar14. Epub 1999 Oct 26. Arthritis Res. 1999. PMID: 11056663 Free PMC article.
-
CNI-1493, an inhibitor of proinflammatory cytokines, retards cartilage destruction in rats with collagen induced arthritis.Ann Rheum Dis. 2005 Mar;64(3):494-6. doi: 10.1136/ard.2004.021550. Ann Rheum Dis. 2005. PMID: 15708902 Free PMC article.
-
Prevention of lethality and suppression of proinflammatory cytokines in experimental septic shock by microencapsulated CNI-1493.J Interferon Cytokine Res. 1999 Oct;19(10):1125-33. doi: 10.1089/107999099313064. J Interferon Cytokine Res. 1999. PMID: 10547152
-
Suppression of TNF-alpha expression, inhibition of Th1 activity, and amelioration of collagen-induced arthritis by rolipram.J Immunol. 1997 Dec 15;159(12):6253-9. J Immunol. 1997. PMID: 9550429
Cited by
-
Citrullinated proteins have increased immunogenicity and arthritogenicity and their presence in arthritic joints correlates with disease severity.Arthritis Res Ther. 2005;7(3):R458-67. doi: 10.1186/ar1697. Epub 2005 Feb 21. Arthritis Res Ther. 2005. PMID: 15899032 Free PMC article.
-
Mitogen activated protein (MAP) kinase signal transduction pathways and novel anti-inflammatory targets.Gut. 2003 Jan;52(1):144-51. doi: 10.1136/gut.52.1.144. Gut. 2003. PMID: 12477778 Free PMC article. Review.
-
Parasite-mediated down-regulation of collagen-induced arthritis (CIA) in DA rats.Clin Exp Immunol. 2000 Dec;122(3):477-83. doi: 10.1046/j.1365-2249.2000.01384.x. Clin Exp Immunol. 2000. PMID: 11122258 Free PMC article.
-
The vagus nerve and nicotinic receptors involve inhibition of HMGB1 release and early pro-inflammatory cytokines function in collagen-induced arthritis.J Clin Immunol. 2010 Mar;30(2):213-20. doi: 10.1007/s10875-009-9346-0. Epub 2009 Nov 5. J Clin Immunol. 2010. PMID: 19890701
-
Suppressive effects of a quinoxaline-analogue (Rob 803) on pathogenic immune mechanisms in collagen-induced arthritis.Clin Exp Immunol. 2008 Apr;152(1):192-9. doi: 10.1111/j.1365-2249.2008.03613.x. Epub 2008 Feb 25. Clin Exp Immunol. 2008. PMID: 18307516 Free PMC article.
References
-
- Trentham DE. Collagen arthritis as a relevant model for rheumatoid arthritis: evidence pro and con. Arthritis Rheum. 1982;25:911–6. - PubMed
-
- Staines NA, Wooley PH. Collagen arthritis—what can it teach us? Br J Rheumatol. 1994;33:798–807. - PubMed
-
- Larsson P, Kleinau S, Holmdahl R, Klareskog L. Homologous type II collagen induced arthritis in rats. Characterization of the disease and demonstration of clinically distinct forms of arthritis in two strains of rats after immunization with the same collagen preparation. Arthritis Rheum. 1990;33:693–701. - PubMed
-
- Cremer MA, Griffiths MM, Terato K, Kang AH. Type XI and II collagen-induced arthritis in rats: characterization of inbred strains of rats for arthritis-susceptibility and immune responsiveness to type XI and II collagen. Autoimmunity. 1995;20:153–61. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

