The stem-loop binding protein (SLBP1) is present in coiled bodies of the Xenopus germinal vesicle
- PMID: 9950690
- PMCID: PMC25182
- DOI: 10.1091/mbc.10.2.487
The stem-loop binding protein (SLBP1) is present in coiled bodies of the Xenopus germinal vesicle
Abstract
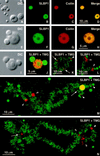
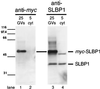
The stem-loop binding protein (SLBP1) binds the 3' stem-loop of histone pre-mRNA and is required for efficient processing of histone transcripts in the nucleus. We examined the localization of SLBP1 in the germinal vesicle of Xenopus laevis oocytes. In spread preparations of germinal vesicle contents, an anti-SLBP1 antibody stained coiled bodies and specific chromosomal loci, including terminal granules, axial granules, and some loops. After injection of myc-tagged SLBP1 transcripts into the oocyte cytoplasm, newly translated myc-SLBP1 protein was detectable in coiled bodies within 4 h and in terminal and axial granules by 8 h. To identify the region(s) of SLBP1 necessary for subnuclear localization, we subcloned various parts of the SLBP1 cDNA and injected transcripts of these into the cytoplasm of oocytes. We determined that 113 amino acids at the carboxy terminus of SLBP1 are sufficient for coiled body localization and that disruption of a previously defined RNA-binding domain did not alter this localization. Coiled bodies also contain the U7 small nuclear ribonucleoprotein particle (snRNP), which participates in cleavage of the 3' end of histone pre-mRNA. The colocalization of SLBP1 and the U7 snRNP in the coiled body suggests coordinated control of their functions, perhaps through a larger histone-processing particle. Some coiled bodies are attached to the lampbrush chromosomes at the histone gene loci, consistent with the view that coiled bodies in the oocyte recruit histone-processing factors to the sites of histone pre-mRNA transcription. The non-histone chromosomal sites at which SLBP1 is found include the genes coding for 5 S rRNA, U1 snRNA, and U2 snRNA, suggesting a wider role for SLBP1 in the biosynthesis of small non-spliced RNAs.
Figures





References
-
- Bond UM, Yario TA, Steitz JA. Multiple processing-defective mutations in a mammalian histone pre-mRNA are suppressed by compensatory changes in U7 RNA both in vivo and in vitro. Genes Dev. 1991;5:1709–1722. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

