Stimulation of myelin basic protein gene transcription by Fyn tyrosine kinase for myelination
- PMID: 9952416
- PMCID: PMC6786019
- DOI: 10.1523/JNEUROSCI.19-04-01393.1999
Stimulation of myelin basic protein gene transcription by Fyn tyrosine kinase for myelination
Abstract
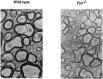
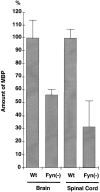
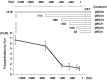
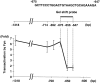
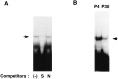
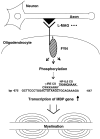
Myelin is synthesized about the time of birth. The Src-family tyrosine kinase Fyn is involved in the initial events of myelination. Fyn is present in myelin-forming cells and is activated through stimulation of cell surface receptors such as large myelin-associated glycoprotein (L-MAG). Here we show that Fyn stimulates transcription of the myelin basic protein (MBP) gene for myelination. MBP is a major component of the myelin membrane. In 4-week-old Fyn-deficient mice, MBP is significantly reduced, and electron microscopic analysis showed that myelination is delayed, compared with wild-type mice. The Fyn-deficient mice had thinner, more irregular myelin than the wild-type. We found that Fyn stimulates the promoter activity of the MBP gene by approximately sevenfold. The region responsible for the transactivation by Fyn is located between nucleotides -675 and -647 with respect to the transcription start site. Proteins binding to this region were found by gel shift study, and the binding activity correlates with Fyn activity during myelination. These results suggest that transactivation of the MBP gene by Fyn is important for myelination.
Figures


References
-
- Asipu A, Blair GE. Regulation of myelin basic protein-encoding gene transcription in rat oligodendrocytes. Gene. 1994;150:227–234. - PubMed
-
- Devine-Beach K, Lashgari MS, Khalili K. Myelin basic protein gene transcription. Identification of proximal and distal cis-acting regulatory elements. J Biol Chem. 1990;265:13830–13835. - PubMed
-
- Haque NS, Buchberg AM, Khalili K. Isolation and characterization of MRF-1, a brain-derived DNA-binding protein with a capacity to regulate expression of myelin basic protein gene. J Biol Chem. 1994;269:31149–31156. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
