Regulation of growth anisotropy in well-watered and water-stressed maize roots. II. Role Of cortical microtubules and cellulose microfibrils
- PMID: 9952465
- PMCID: PMC32146
- DOI: 10.1104/pp.119.2.681
Regulation of growth anisotropy in well-watered and water-stressed maize roots. II. Role Of cortical microtubules and cellulose microfibrils
Abstract
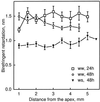
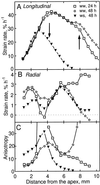
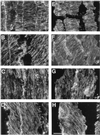
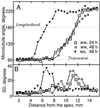
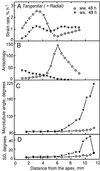
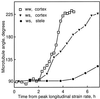
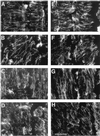
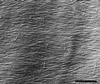
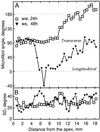
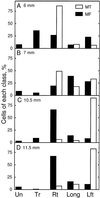
We tested the hypothesis that the degree of anisotropic expansion of plant tissues is controlled by the degree of alignment of cortical microtubules or cellulose microfibrils. Previously, for the primary root of maize (Zea mays L.), we quantified spatial profiles of expansion rate in length, radius, and circumference and the degree of growth anisotropy separately for the stele and cortex, as roots became thinner with time from germination or in response to low water potential (B.M. Liang, A.M. Dennings, R.E. Sharp, T.I. Baskin [1997] Plant Physiol 115:101-111). Here, for the same material, we quantified microtubule alignment with indirect immunofluorescence microscopy and microfibril alignment throughout the cell wall with polarized-light microscopy and from the innermost cell wall layer with electron microscopy. Throughout much of the growth zone, mean orientations of microtubules and microfibrils were transverse, consistent with their parallel alignment specifying the direction of maximal expansion rate (i.e. elongation). However, where microtubule alignment became helical, microfibrils often made helices of opposite handedness, showing that parallelism between these elements was not required for helical orientations. Finally, contrary to the hypothesis, the degree of growth anisotropy was not correlated with the degree of alignment of either microtubules or microfibrils. The mechanisms plants use to specify radial and tangential expansion rates remain uncharacterized.
Figures
References
-
- Abeysekera RM, McCully M. The epidermal surface of the maize root tip. I. Development in normal roots. New Phytol. 1993a;125:413–430. - PubMed
-
- Abeysekera RM, McCully M. The epidermal surface of the maize root tip. II. Abnormalities in a mutant which grows crookedly through soil. New Phytol. 1993b;125:801–811. - PubMed
-
- Abeysekera RM, McCully M. The epidermal surface of the maize root tip. III. Isolation of the surface and characterization of some of its structural and mechanical properties. New Phytol. 1994;127:321–333. - PubMed
-
- Akashi T, Shibaoka H. Effects of gibberellin on the arrangement and the cold stability of cortical microtubules in epidermal cells of pea internodes. Plant Cell Physiol. 1987;28:339–348.
-
- Arioli T, Peng L, Betzner AS, Burn J, Wittke W, Herth W, Camilleri C, Höfte H, Plazinski J, Birch R and others. Molecular analysis of cellulose biosynthesis in Arabidopsis. Science. 1998;179:717–720. - PubMed
LinkOut - more resources
Full Text Sources

