Mammalian homologue of the Caenorhabditis elegans UNC-76 protein involved in axonal outgrowth is a protein kinase C zeta-interacting protein
- PMID: 9971736
- PMCID: PMC2132904
- DOI: 10.1083/jcb.144.3.403
Mammalian homologue of the Caenorhabditis elegans UNC-76 protein involved in axonal outgrowth is a protein kinase C zeta-interacting protein
Abstract
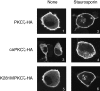
By the yeast two-hybrid screening of a rat brain cDNA library with the regulatory domain of protein kinase C zeta (PKCzeta) as a bait, we have cloned a gene coding for a novel PKCzeta-interacting protein homologous to the Caenorhabditis elegans UNC-76 protein involved in axonal outgrowth and fasciculation. The protein designated FEZ1 (fasciculation and elongation protein zeta-1) consisting of 393 amino acid residues shows a high Asp/Glu content and contains several regions predicted to form amphipathic helices. Northern blot analysis has revealed that FEZ1 mRNA is abundantly expressed in adult rat brain and throughout the developmental stages of mouse embryo. By the yeast two-hybrid assay with various deletion mutants of PKC, FEZ1 was shown to interact with the NH2-terminal variable region (V1) of PKCzeta and weakly with that of PKCepsilon. In the COS-7 cells coexpressing FEZ1 and PKCzeta, FEZ1 was present mainly in the plasma membrane, associating with PKCzeta and being phosphorylated. These results indicate that FEZ1 is a novel substrate of PKCzeta. When the constitutively active mutant of PKCzeta was used, FEZ1 was found in the cytoplasm of COS-7 cells. Upon treatment of the cells with a PKC inhibitor, staurosporin, FEZ1 was translocated from the cytoplasm to the plasma membrane, suggesting that the cytoplasmic translocation of FEZ1 is directly regulated by the PKCzeta activity. Although expression of FEZ1 alone had no effect on PC12 cells, coexpression of FEZ1 and constitutively active PKCzeta stimulated the neuronal differentiation of PC12 cells. Combined with the recent finding that a human FEZ1 protein is able to complement the function of UNC-76 necessary for normal axonal bundling and elongation within axon bundles in the nematode, these results suggest that FEZ1 plays a crucial role in the axon guidance machinery in mammals by interacting with PKCzeta.
Figures





References
-
- Colavita A, Krishna S, Zheng H, Padgett RW, Culotti JG. Pioneer axon guidance by UNC-129, a C. elegansTGF-β. Science. 1998;281:706–709. - PubMed
-
- Desai C, Garriga G, McIntire SL, Horvitz HR. A genetic pathway for the development of the Caenorhabditis elegansHSN motor neurons. Nature. 1988;336:638–646. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

