Generation of neutralizing human monoclonal antibodies against parvovirus B19 proteins
- PMID: 9971777
- PMCID: PMC104439
- DOI: 10.1128/JVI.73.3.1974-1979.1999
Generation of neutralizing human monoclonal antibodies against parvovirus B19 proteins
Abstract
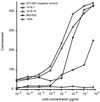
Infections caused by human parvovirus B19 are known to be controlled mainly by neutralizing antibodies. To analyze the immune reaction against parvovirus B19 proteins, four cell lines secreting human immunoglobulin G monoclonal antibodies (MAbs) were generated from two healthy donors and one human immunodeficiency virus type 1-seropositive individual with high serum titers against parvovirus. One MAb is specific for nonstructural protein NS1 (MAb 1424), two MAbs are specific for the unique region of minor capsid protein VP1 (MAbs 1418-1 and 1418-16), and one MAb is directed to major capsid protein VP2 (MAb 860-55D). Two MAbs, 1418-1 and 1418-16, which were generated from the same individual have identity in the cDNA sequences encoding the variable domains, with the exception of four base pairs resulting in only one amino acid change in the light chain. The NS1- and VP1-specific MAbs interact with linear epitopes, whereas the recognized epitope in VP2 is conformational. The MAbs specific for the structural proteins display strong virus-neutralizing activity. The VP1- and VP2-specific MAbs have the capacity to neutralize 50% of infectious parvovirus B19 in vitro at 0.08 and 0.73 microgram/ml, respectively, demonstrating the importance of such antibodies in the clearance of B19 viremia. The NS1-specific MAb mediated weak neutralizing activity and required 47.7 micrograms/ml for 50% neutralization. The human MAbs with potent neutralizing activity could be used for immunotherapy of chronically B19 virus-infected individuals and acutely infected pregnant women. Furthermore, the knowledge gained regarding epitopes which induce strongly neutralizing antibodies may be important for vaccine development.
Figures



References
-
- Anderson M J, Jones S E, Fisher H S, Lewis E, Hall S M, Bartlett C L, Cohen B J, Mortimer P P, Pereira M S. Human parvovirus, the cause of erythema infectiosum (fifth disease)? Lancet. 1983;i:1378. - PubMed
-
- Arakelov S, Gorny M K, Williams C, Riggin C H, Brady F, Collett M S, Zolla-Pazner S. Generation of neutralizing anti-B19 parvovirus human monoclonal antibodies from patients infected with human immunodeficiency virus. J Infect Dis. 1993;168:580–585. - PubMed
-
- Brown K E, Green S W, O’Sullivan M G, Young N S. Cloning and sequencing of the simian parvovirus genome. Virology. 1995;210:314–322. - PubMed
-
- Brown T, Anand A, Ritchie L D, Clewley J P, Reid T M. Intrauterine parvovirus infection associated with hydrops fetalis. Lancet. 1984;ii:1033–1034. . (Letter.) - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

