Probing nucleosome function: a highly versatile library of synthetic histone H3 and H4 mutants
- PMID: 18805098
- PMCID: PMC2701395
- DOI: 10.1016/j.cell.2008.07.019
Probing nucleosome function: a highly versatile library of synthetic histone H3 and H4 mutants
Abstract
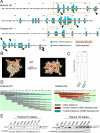
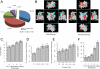
Nucleosome structural integrity underlies the regulation of DNA metabolism and transcription. Using a synthetic approach, a versatile library of 486 systematic histone H3 and H4 substitution and deletion mutants that probes the contribution of each residue to nucleosome function was generated in Saccharomyces cerevisiae. We probed fitness contributions of each residue to perturbations of chromosome integrity and transcription, mapping global patterns of chemical sensitivities and requirements for transcriptional silencing onto the nucleosome surface. Each histone mutant was tagged with unique molecular barcodes, facilitating identification of histone mutant pools through barcode amplification, labeling, and TAG microarray hybridization. Barcodes were used to score complex phenotypes such as competitive fitness in a chemostat, DNA repair proficiency, and synthetic genetic interactions, revealing new functions for distinct histone residues and new interdependencies among nucleosome components and their modifiers.
Figures




References
-
- Adkins MW, Carson JJ, English CM, Ramey CJ, Tyler JK. The histone chaperone anti-silencing function 1 stimulates the acetylation of newly synthesized histone H3 in S-phase. J. Biol. Chem. 2007;282:1334–1340. - PubMed
-
- Berger SL. The complex language of chromatin regulation during transcription. Nature. 2007;447:407–412. - PubMed
-
- Bryk M, Banerjee M, Murphy M, Knudsen KE, Garfinkel DJ, Curcio MJ. Transcriptional silencing of Ty1 elements in the RDN1 locus of yeast. Genes Dev. 1997;11:255–269. - PubMed
-
- Bryk M, Briggs SD, Strahl BD, Curcio MJ, Allis CD, Winston F. Evidence that Set1, a factor required for methylation of histone H3, regulates rDNA silencing in S. cerevisiae by a Sir2-independent mechanism. Curr. Biol. 2002;12:165–170. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

