A suberized exodermis is required for tomato drought tolerance
- PMID: 38168610
- PMCID: PMC10808073
- DOI: 10.1038/s41477-023-01567-x
A suberized exodermis is required for tomato drought tolerance
Abstract
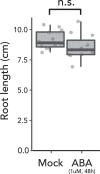
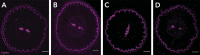
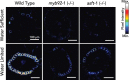
Plant roots integrate environmental signals with development using exquisite spatiotemporal control. This is apparent in the deposition of suberin, an apoplastic diffusion barrier, which regulates flow of water, solutes and gases, and is environmentally plastic. Suberin is considered a hallmark of endodermal differentiation but is absent in the tomato endodermis. Instead, suberin is present in the exodermis, a cell type that is absent in the model organism Arabidopsis thaliana. Here we demonstrate that the suberin regulatory network has the same parts driving suberin production in the tomato exodermis and the Arabidopsis endodermis. Despite this co-option of network components, the network has undergone rewiring to drive distinct spatial expression and with distinct contributions of specific genes. Functional genetic analyses of the tomato MYB92 transcription factor and ASFT enzyme demonstrate the importance of exodermal suberin for a plant water-deficit response and that the exodermal barrier serves an equivalent function to that of the endodermis and can act in its place.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures







References
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
- 55108506/Howard Hughes Medical Institute (HHMI)
- PGRP IOS-1856749/National Science Foundation (NSF)
- PGRP IOS-211980/National Science Foundation (NSF)
- MCB-2218234/National Science Foundation (NSF)
- PRFB IOS-1907008/National Science Foundation (NSF)
- DGE-1922642/National Science Foundation (NSF)
- RI 790057/EC | EU Framework Programme for Research and Innovation H2020 | H2020 Priority Excellent Science | H2020 Marie Sklodowska-Curie Actions (H2020 Excellent Science - Marie Sklodowska-Curie Actions)
- FI-570-2018/United States - Israel Binational Agricultural Research and Development Fund (BARD)
- #RGP0067/Human Frontier Science Program (HFSP)
- DE-SC0020358/DOE | Advanced Research Projects Agency - Energy (Advanced Research Projects Agency - Energy - U.S. Department of Energy)
- 1026477/DOE | Advanced Research Projects Agency - Energy (Advanced Research Projects Agency - Energy - U.S. Department of Energy)
- ALTF 1046-2015/European Molecular Biology Organization (EMBO)
- 1026477/United States Department of Agriculture | National Institute of Food and Agriculture (NIFA)
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

