Heterosexual Transmission of Subtype C HIV-1 Selects Consensus-Like Variants without Increased Replicative Capacity or Interferon-α Resistance
- PMID: 26378795
- PMCID: PMC4574710
- DOI: 10.1371/journal.ppat.1005154
Heterosexual Transmission of Subtype C HIV-1 Selects Consensus-Like Variants without Increased Replicative Capacity or Interferon-α Resistance
Abstract
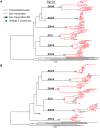
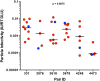
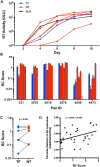
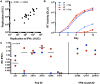
Heterosexual transmission of HIV-1 is characterized by a genetic bottleneck that selects a single viral variant, the transmitted/founder (TF), during most transmission events. To assess viral characteristics influencing HIV-1 transmission, we sequenced 167 near full-length viral genomes and generated 40 infectious molecular clones (IMC) including TF variants and multiple non-transmitted (NT) HIV-1 subtype C variants from six linked heterosexual transmission pairs near the time of transmission. Consensus-like genomes sensitive to donor antibodies were selected for during transmission in these six transmission pairs. However, TF variants did not demonstrate increased viral fitness in terms of particle infectivity or viral replicative capacity in activated peripheral blood mononuclear cells (PBMC) and monocyte-derived dendritic cells (MDDC). In addition, resistance of the TF variant to the antiviral effects of interferon-α (IFN-α) was not significantly different from that of non-transmitted variants from the same transmission pair. Thus neither in vitro viral replicative capacity nor IFN-α resistance discriminated the transmission potential of viruses in the quasispecies of these chronically infected individuals. However, our findings support the hypothesis that within-host evolution of HIV-1 in response to adaptive immune responses reduces viral transmission potential.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Derdeyn CA, Decker JM, Bibollet-Ruche F, Mokili JL, Muldoon M, Denham SA, et al. Envelope-constrained neutralization-sensitive HIV-1 after heterosexual transmission. Science. 2004. March 26;303(5666):2019–22. - PubMed
-
- Haaland RE, Hawkins PA, Salazar-Gonzalez J, Johnson A, Tichacek A, Karita E, et al. Inflammatory Genital Infections Mitigate a Severe Genetic Bottleneck in Heterosexual Transmission of Subtype A and C HIV-1. Trkola A, editor. PLoS Pathog. 2009. January 23;5(1):e1000274 10.1371/journal.ppat.1000274 - DOI - PMC - PubMed
-
- Salazar-Gonzalez JF, Salazar MG, Keele BF, Learn GH, Giorgi EE, Li H, et al. Genetic identity, biological phenotype, and evolutionary pathways of transmitted/founder viruses in acute and early HIV-1 infection. Journal of Experimental Medicine. 2009. June 8;206(6):1273–89. 10.1084/jem.20090378 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

