Glycopyrronium once-daily significantly improves lung function and health status when combined with salmeterol/fluticasone in patients with COPD: the GLISTEN study, a randomised controlled trial
- PMID: 25841237
- PMCID: PMC4453631
- DOI: 10.1136/thoraxjnl-2014-206670
Glycopyrronium once-daily significantly improves lung function and health status when combined with salmeterol/fluticasone in patients with COPD: the GLISTEN study, a randomised controlled trial
Abstract
Background: The optimal use of various therapeutic combinations for moderate/severe chronic obstructive pulmonary disease (COPD) is unclear. The GLISTEN trial compared the efficacy of two long-acting anti-muscarinic antagonists (LAMA), when combined with an inhaled corticosteroid (ICS) and a long-acting β2 agonist (LABA).
Methods: This randomised, blinded, placebo-controlled trial in moderate/severe COPD patients compared once-daily glycopyrronium (GLY) 50 µg, once-daily tiotropium (TIO) 18 µg or placebo (PLA), when combined with salmeterol/fluticasone propionate (SAL/FP) 50/500 µg twice daily. The primary objective was to determine the non-inferiority of GLY+SAL/FP versus TIO+SAL/FP on trough FEV1 after 12 weeks. An important secondary objective was whether addition of GLY to SAL/FP was better than SAL/FP alone.
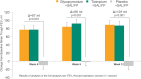
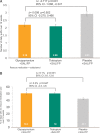
Results: 773 patients (mean FEV1 57.2% predicted) were randomised; 84.9% completed the trial. At week 12, GLY+SAL/FP demonstrated non-inferiority to TIO+SAL/FP for trough FEV1: least square mean treatment difference (LSMdiff) -7 mL (SE 17.4) with a lower limit for non-inferiority of -60 mL. There was significant increase in week 12 trough FEV1 with GLY+SAL/FP versus PLA+SAL/FP (LSMdiff 101 mL, p<0.001). At 12 weeks, GLY+SAL/FP produced significant improvement in St George's Respiratory Questionnaire total score versus PLA+SAL/FP (LSMdiff -2.154, p=0.02). GLY+SAL/FP demonstrated significant rescue medication reduction versus PLA+SAL/FP (LSMdiff -0.72 puffs/day, p<0.001). Serious adverse events were similar for GLY+SAL/FP, TIO+SAL/FP and PLA+SAL/FP with an incidence of 5.8%, 8.5% and 5.8%, respectively.
Conclusions: GLY+SAL/FP showed comparable improvements in lung function, health status and rescue medication to TIO+SAL/FP. Importantly, addition of GLY to SAL/FP demonstrated significant improvements in lung function, health status and rescue medication compared to SAL/FP.
Trial registration number: NCT01513460.
Keywords: COPD Pharmacology.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://group.bmj.com/group/rights-licensing/permissions.
Figures



Comment in
-
Triple inhaler therapy for COPD.Thorax. 2015 Oct;70(10):991. doi: 10.1136/thoraxjnl-2015-207388. Epub 2015 Jun 19. Thorax. 2015. PMID: 26092923 No abstract available.
References
-
- The Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD), 2014. http://www.goldcopd.org.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
