On the structure of the stator of the mitochondrial ATP synthase
- PMID: 16791136
- PMCID: PMC1500866
- DOI: 10.1038/sj.emboj.7601177
On the structure of the stator of the mitochondrial ATP synthase
Abstract
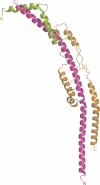
The structure of most of the peripheral stalk, or stator, of the F-ATPase from bovine mitochondria, determined at 2.8 A resolution, contains residues 79-183, 3-123 and 5-70 of subunits b, d and F6, respectively. It consists of a continuous curved alpha-helix about 160 A long in the single b-subunit, augmented by the predominantly alpha-helical d- and F6-subunits. The structure occupies most of the peripheral stalk in a low-resolution structure of the F-ATPase. The long helix in subunit b extends from near to the top of the F1 domain to the surface of the membrane domain, and it probably continues unbroken across the membrane. Its uppermost region interacts with the oligomycin sensitivity conferral protein, bound to the N-terminal region of one alpha-subunit in the F1 domain. Various features suggest that the peripheral stalk is probably rigid rather than resembling a flexible rope. It remains unclear whether the transient storage of energy required by the rotary mechanism takes place in the central stalk or in the peripheral stalk or in both domains.
Figures


References
-
- Abrahams JP, Leslie AGW (1996) Methods used in the structure determination of bovine mitochondrial F1 ATPase. Acta Crystallogr D 52: 30–42 - PubMed
-
- Abrahams JP, Leslie AGW, Lutter R, Walker JE (1994) Structure at 2.8 Å resolution of F1-ATPase from bovine heart mitochondria. Nature 370: 621–628 - PubMed
-
- Böttcher B, Schwarz L, Gra̋ber P (1998) Direct indication for the existence of a double stalk in CFoF1. J Mol Biol 281: 757–762 - PubMed
-
- Boyer PD (1998) ATP synthase—past and future. Biochim Biophys Acta 1365: 3–9 - PubMed
-
- Bricogne G, Vonrhein C, Flensburg C, Schiltz M, Paciorek W (2003) Generation, representation and flow of phase information in structure determination: recent developments in and around SHARP 2.0. Acta Crystallogr D 59: 2023–2030 - PubMed
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

