Specificity and reactivity in menaquinone biosynthesis: the structure of Escherichia coli MenD (2-succinyl-5-enolpyruvyl-6-hydroxy-3-cyclohexadiene-1-carboxylate synthase)
- PMID: 18983854
- PMCID: PMC3656419
- DOI: 10.1016/j.jmb.2008.10.048
Specificity and reactivity in menaquinone biosynthesis: the structure of Escherichia coli MenD (2-succinyl-5-enolpyruvyl-6-hydroxy-3-cyclohexadiene-1-carboxylate synthase)
Abstract
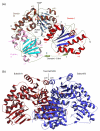
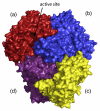
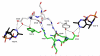
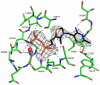
The thiamine diphosphate (ThDP) and metal-ion-dependent enzyme 2-succinyl-5-enolpyruvyl-6-hydroxy-3-cyclohexadiene-1-carboxylate synthase, or MenD, catalyze the Stetter-like conjugate addition of alpha-ketoglutarate with isochorismate to release 2-succinyl-5-enolpyruvyl-6-hydroxy-3-cyclohexadiene-1-carboxylate and carbon dioxide. This reaction represents the first committed step for biosynthesis of menaquinone, or vitamin K2, a key cofactor for electron transport in bacteria and a metabolite for posttranslational modification of proteins in mammals. The medium-resolution structure of MenD from Escherichia coli (EcMenD) in complex with its cofactor and Mn2+ has been determined in two related hexagonal crystal forms. The subunit displays the typical three-domain structure observed for ThDP-dependent enzymes in which two of the domains bind and force the cofactor into a configuration that supports formation of a reactive ylide. The structures reveal a stable dimer-of-dimers association in agreement with gel filtration and analytical ultracentrifugation studies and confirm the classification of MenD in the pyruvate oxidase family of ThDP-dependent enzymes. The active site, created by contributions from a pair of subunits, is highly basic with a pronounced hydrophobic patch. These features, formed by highly conserved amino acids, match well to the chemical properties of the substrates. A model of the covalent intermediate formed after reaction with the first substrate alpha-ketoglutarate and with the second substrate isochorismate positioned to accept nucleophilic attack has been prepared. This, in addition to structural and sequence comparisons with putative MenD orthologues, provides insight into the specificity and reactivity of MenD and allows a two-stage reaction mechanism to be proposed.
Figures




References
-
- Meganathan R. Biosynthesis of the isoprenoid quinones menaquinone (vitamin K2) and ubiquinone (coenzyme Q) In: Neidhardt FC, Curtiss R, Ingraham JL, Lin ECC, Low KB, Magasanik B, Reznikoff WS, Riley M, Schaechter M, Umbarger HE, editors. Escherichia coli and Salmonella: Cellular and Molecular Biology. American Society for Microbiology; Washington, DC: 1996. pp. 642–656.
-
- Meganathan R. Biosynthesis of menaquinone (vitamin K2) and ubiquinone (coenzyme Q): a perspective on enzymatic mechanisms. Vitam. Horm. 2001;61:173–218. - PubMed
-
- Soballe B, Poole RK. Microbial ubiquinones: multiple roles in respiration, gene regulation and oxidative stress management. Microbiology. 1999;145:1817–1830. - PubMed
-
- Bekker M, Kramer G, Hartog AF, Wagner MJ, de Koster CG, Hellingwerf KJ, de Mattos MJ. Changes in the redox state and composition of the quinone pool of Escherichia coli during aerobic batch-culture growth. Microbiology. 2007;153:1974–1980. - PubMed
-
- Bandyopadhyay PK. Vitamin K-dependent gamma-glutamylcarboxylation: an ancient posttranslational modification. Vitam. Horm. 2008;78:157–184. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

