Trapping of palindromic ligands within native transthyretin prevents amyloid formation
- PMID: 21059958
- PMCID: PMC2996680
- DOI: 10.1073/pnas.1008255107
Trapping of palindromic ligands within native transthyretin prevents amyloid formation
Abstract
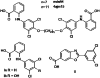
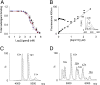
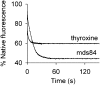
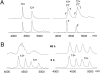
Transthyretin (TTR) amyloidosis is a fatal disease for which new therapeutic approaches are urgently needed. We have designed two palindromic ligands, 2,2'-(4,4'-(heptane-1,7-diylbis(oxy))bis(3,5-dichloro-4,1-phenylene)) bis(azanediyl)dibenzoic acid (mds84) and 2,2'-(4,4'-(undecane-1,11-diylbis(oxy))bis(3,5-dichloro-4,1-phenylene)) bis(azanediyl)dibenzoic acid (4ajm15), that are rapidly bound by native wild-type TTR in whole serum and even more avidly by amyloidogenic TTR variants. One to one stoichiometry, demonstrable in solution and by MS, was confirmed by X-ray crystallographic analysis showing simultaneous occupation of both T4 binding sites in each tetrameric TTR molecule by the pair of ligand head groups. Ligand binding by native TTR was irreversible under physiological conditions, and it stabilized the tetrameric assembly and inhibited amyloidogenic aggregation more potently than other known ligands. These superstabilizers are orally bioavailable and exhibit low inhibitory activity against cyclooxygenase (COX). They offer a promising platform for development of drugs to treat and prevent TTR amyloidosis.
Conflict of interest statement
Conflict of interest statement: S.E.K., V.B., A.J.M., M.D.S., S.V.L., S.P.W., and M.B.P. are the inventors on patent application WO 2009/040405 A1 on 4ajm15 and mds84, which is owned by Pentraxin Therapeutics Ltd, a University College London spinout company in which S.P.W. and M.B.P. have shares.
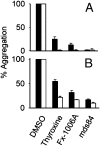
Figures

References
-
- Cornwell GG, 3rd, Murdoch WL, Kyle RA, Westermark P, Pitkänen P. Frequency and distribution of senile cardiovascular amyloid. A clinicopathologic correlation. Am J Med. 1983;75:618–623. - PubMed
-
- Jacobson DR, et al. Variant-sequence transthyretin (isoleucine 122) in late-onset cardiac amyloidosis in black Americans. N Engl J Med. 1997;336:466–473. - PubMed
-
- Saraiva MJ. Hereditary transthyretin amyloidosis: Molecular basis and therapeutical strategies. Expert Rev Mol Med. 2002;4:1–11. - PubMed
-
- Holmgren G, et al. Biochemical effect of liver transplantation in two Swedish patients with familial amyloidotic polyneuropathy (FAP-met30) Clin Genet. 1991;40:242–246. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

