Influence of pharmacogenetics on indinavir disposition and short-term response in HIV patients initiating HAART
- PMID: 19440701
- PMCID: PMC2908290
- DOI: 10.1007/s00228-009-0660-5
Influence of pharmacogenetics on indinavir disposition and short-term response in HIV patients initiating HAART
Abstract
Aims: To assess the relationship between genetic polymorphisms and indinavir pharmacokinetic variability and to study the link between concentrations and short-term response or metabolic safety.
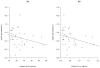
Methods: Forty protease inhibitor-naive patients initiating highly active antiretroviral therapy (HAART) including indinavir/ritonavir and enrolled in the COPHAR 2-ANRS 111 trial were studied. At week 2, four blood samples were taken before and up to 6 h following drug intake. A population pharmacokinetic analysis was performed using the stochastic approximation expectation maximization (SAEM) algorithm implemented in MONOLIX software. The area under the concentration-time curve (AUC) and maximum (C(max)) and trough concentrations (C(trough)) of indinavir were derived from the population model and tested for their correlation with short-term viral response and safety measurements, while for ritonavir, these same three parameters were tested for their correlation with short-term biochemical safety
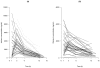
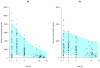
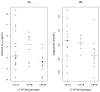
Results: A one-compartment model with first-order absorption and elimination best described both indinavir and ritonavir concentrations. For indinavir, the estimated clearance and volume of distribution were 22.2 L/h and 97.3 L, respectively. The eight patients with the *1B/*1B genotype for the CYP3A4 gene showed a 70% decrease in absorption compared to those with the *1A/*1B or *1A/*1A genotypes (0.5 vs. 2.1, P = 0.04, likelihood ratio test by permutation). The indinavir AUC and C(trough) were positively correlated with the decrease in human immunodeficiency virus RNA between week 0 and week 2 (r = 0.4, P = 0.03 and r = -0.4, P = 0.03, respectively). Patients with the *1B/*1B genotype also had a significantly lower indinavir C(max) (median 3.6, range 2.1-5.2 ng/mL) than those with the *1A/*1B or *1A/*1A genotypes (median 4.4, range 2.2-8.3 ng/mL) (P = 0.04) and a lower increase in triglycerides during the first 4 weeks of treatment (median 0.1, range -0.7 to 1.4 vs. median 0.6, range -0.5 to 1.7 mmol/L, respectively; P = 0.02). For ritonavir, the estimated clearance and volume of distribution were 8.3 L/h and 60.7 L, respectively, and concentrations were not found to be correlated to biochemical safety. Indinavir and ritonavir absorption rate constants were found to be correlated, as well as their apparent volumes of distribution and clearances, indicating correlated bioavailability of the two drugs.
Conclusion: The CYP3A4*1B polymorphism was found to influence the pharmacokinetics of indinavir and, to some extent, the biochemical safety of indinavir.
Figures
References
-
- Collin F, Chêne G, Retout S, et al. Indinavir trough concentration as a determinant of early nephrolithiasis in HIV-1-infected adults. Ther Drug Monit. 2007;29:164–70. - PubMed
-
- Flexner C. HIV-protease inhibitors. N Engl J Med. 1998;338:1281–92. - PubMed
-
- Von Moltke LL, Greenblatt DJ, Grassi JM, et al. Protease inhibitors as inhibitors of human cytochromes P450: high risk associated with ritonavir. J Clin Pharmacol. 1998;38:106–11. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

